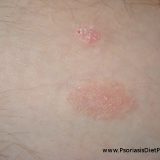
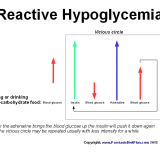
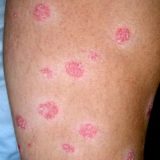
LDN for Psoriasis: Good Treatment But Still Not a Cure
Low Dose Naltrexone (LDN) is a popular alternative treatment not only for psoriasis. Still the most people do not get it and are clueless about the reasons why and how Naltrexone in low dosages works for so many chronic inflammatory and degenerative diseases. Many doctors, chiropractors and naturopaths agree that LDN is quite an effective and relatively very safe treatment for many diseases. Since I am absolutely sure that psoriasis is an Autonomic Nervous System dysfunction rather than autoimmune disease…