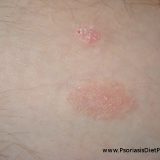
Psoriatics: Saccharomyces Boulardii Is Dangerous!
Even though Saccharomyces boulardii is one of the most popular digestive and SIBO supplements it is nowhere near as safe and perhaps also effective as you might think. Yes, I know there is another article on this blog – “Probiotics are Useless, Saccharomyces Boulardii as SIBO Cure”. I would not oppose the information stated in that blog post which was written in order to explain why bacterial probiotics may not be the best way how to cope with digestive or…